How real-world data and analytics help accelerate clinical trials

Shweta Maniar
Global Director, Life Science Strategy & Solutions, Google Cloud
How real-world data and analytics platforms help accelerate clinical trials for life sciences.
The collective response to COVID-19 was unprecedented, as heroes on the front lines and behind the scenes joined forces against a pandemic that threatened people in every corner of the world.
Discovering, developing, manufacturing, and distributing vaccines and therapies in record time required stunning measures: allocating billions in public funding, running clinical trials in parallel, expediting regulatory approvals, pre-ramping manufacturing, and setting up infrastructure to supply products. Thousands of communities received shots in arms, therapies, and equipment requiring regulatory oversight within months instead of the typical 5 to 15 years.
Did Operation Warp Speed and other international efforts set a new precedent for expediting future pharmaceutical development? Absolutely. And yet—the remarkable breakthroughs of the past two years have presented the life sciences industry with a conundrum it will have to confront for many more years to come.
The public will likely now greet life sciences companies with less patience for decade-long drug development cycles and more demands for retail-like transparency. Because the pharmaceutical industry needs to update its processes, meeting these expectations won’t be as simple as replicating the COVID-19 vaccine model. How, then, might the healthcare industry accelerate bringing new life saving treatments to the market safely and more quickly in the absence of a public emergency?
The cost of not loving your real-world data platform
Reducing development time and cost for new vaccines, treatments, and devices has been—and remains—the apex achievement for life sciences companies and the public they serve. Without sacrificing patient safety, companies can accelerate clinical trials and increase patient participation by making strategic use of real-world data, insights, and evidence. More data from more people generates more knowledge about how diseases work and how to defeat them, and also how specific populations respond to specific treatments.
Gathering and analyzing real-world data is not a simple task, though, requiring careful orchestration of many moving parts. As a result, life sciences companies have invested heavily in technology to help manage their inputs and compliance.
Few of them, however, are happy with the technology they currently use for garnering timely, actionable insights.
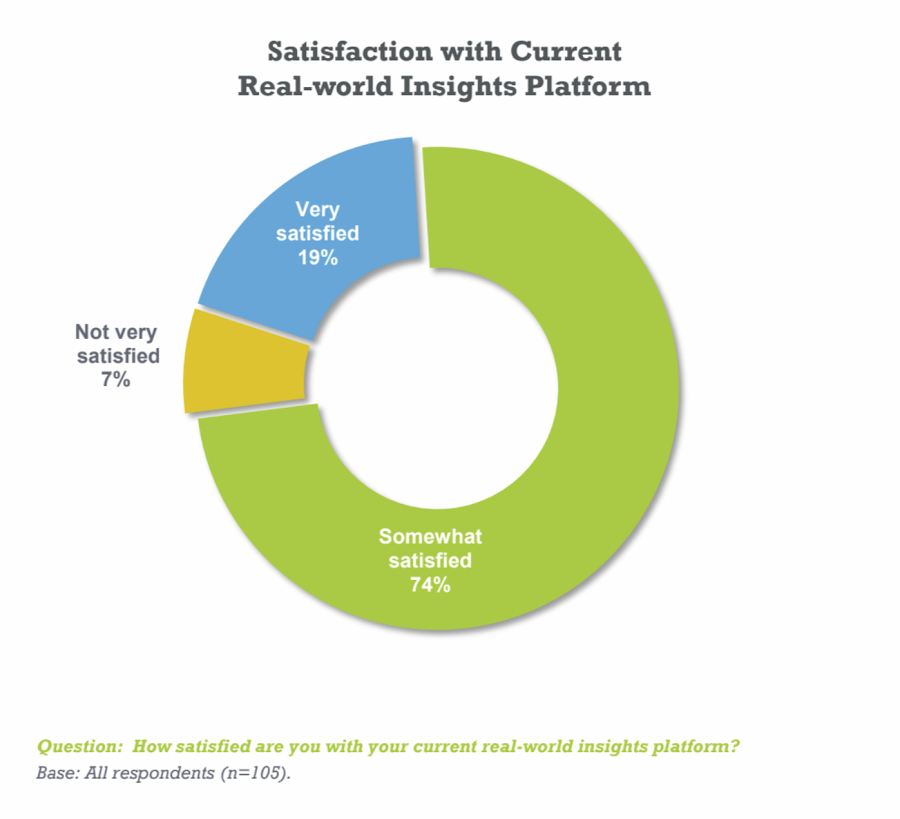
According to a recent survey conducted by Pharma Intelligence with Google Cloud, only 19 percent of respondents using real-world data are “very satisfied” with their technology platform. Typical limitations cited include cost, speed, capacity, expertise requirements, and lack of customizability.

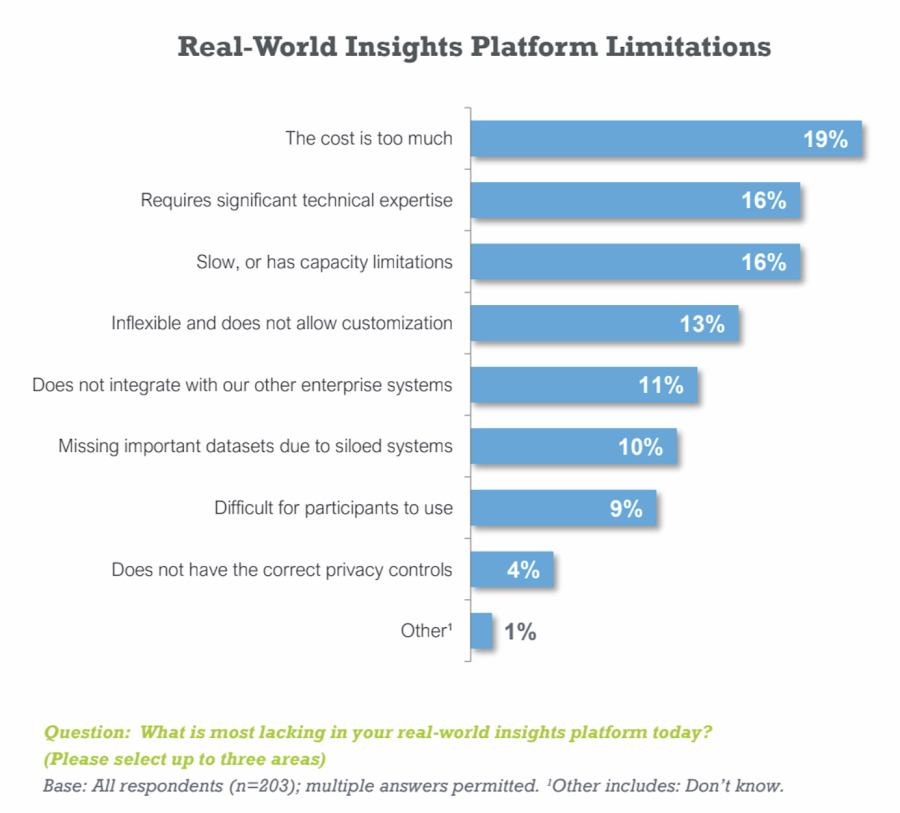

Companies wrestling with these constraints are understandably frustrated by constantly needing to reinvest in technology to improve functionality and keep pace with regulatory changes. But what if a lower-cost, pharma-ready solution with near infinite capacity was available—and deployable in one click? And what if the U.S. Food and Drug Administration had already released it as an open source platform that clinical researchers can use today?
A new speed to clinical trials
The cloud computing era has brought researchers a host of cross-platform, open-source tools that enable a new level of efficiency, collaboration, and security, as well as faster time-to-insight from most any type of data. Many leading life sciences enterprises have turned to Google Cloud’s Healthcare and Life Sciences solutions to provide them with healthcare-specific functionality. These tools help ingest, aggregate, and de-identify any type of healthcare data in its original format (such as DICOM or FHIR) to make data useful throughout drug development, from discovery to commercialization.
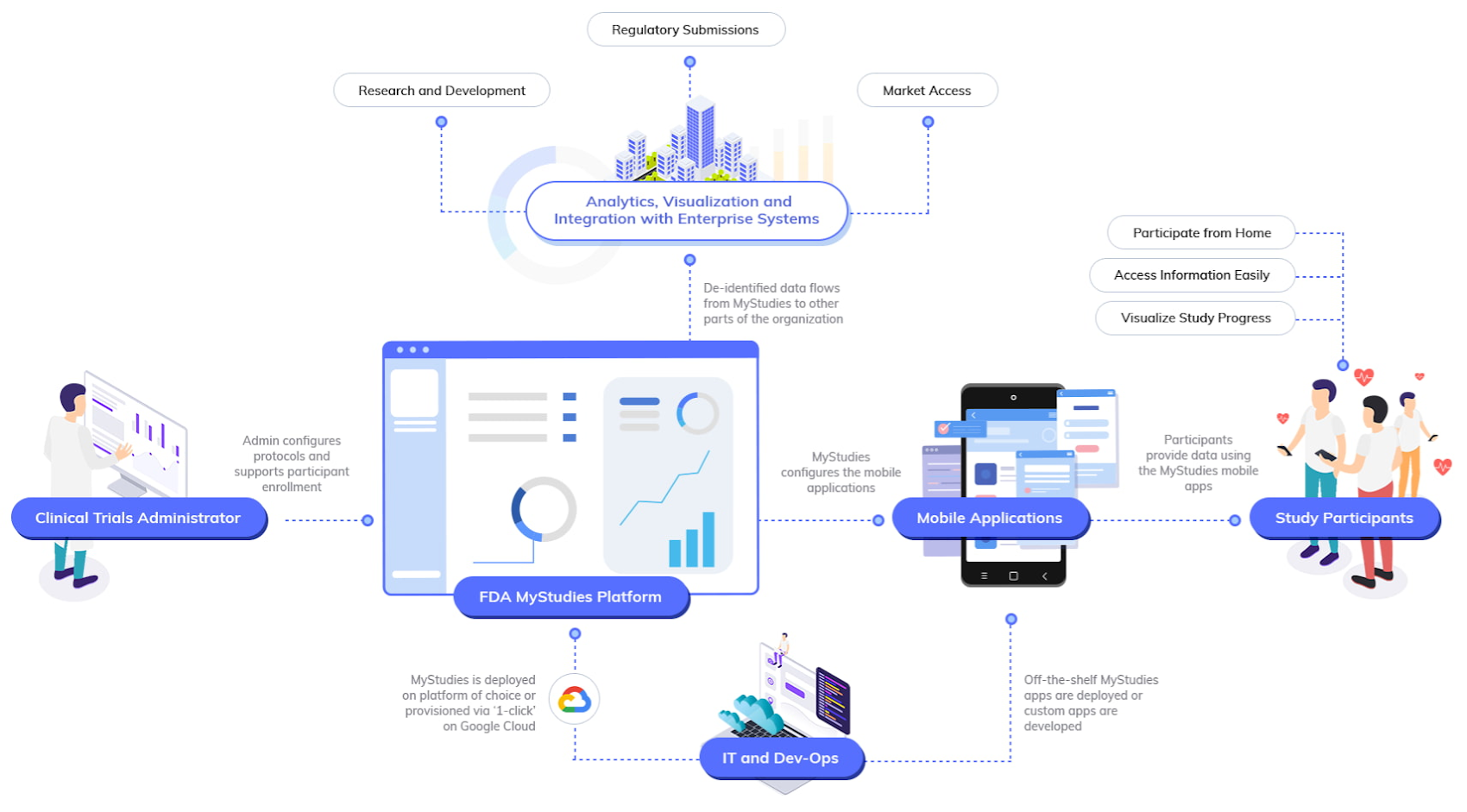
One of these solutions, our Real-World Insights platform, is based on FDA MyStudies and gives clinical trial sponsors instant access to necessary digital components for designing, planning, and executing studies. Its pre-built architectural components include study builder, study datastore, participant datastore, response datastore, and participant manager.


Because the Real-World Insights platform supports healthcare industry data interoperability standards, it can help trial sponsors more easily harness and organize data from a variety of sources. For example, they can build personalized, consumer-like apps that will inspire increased participation from a wider pool of patient candidates and make it easier for researchers to collect, manage, and analyze trial data.
The solution enables researchers to ask new questions, get answers more quickly, and work more collaboratively, thus reducing timelines through automation. Institutions can take advantage of virtually unlimited storage and processing capacity to scale to more ambitious projects and generate actionable, real-time insights from any data source, all while staying within budget.
Top research centers have already made the shift to storing and analyzing data on Google Cloud, finding the Real-World Insights platform faster and more cost effective than downloading and storing data on their own servers.
Using real-world data to reduce development time and cost
Although we’re still in the early stages of using cloud computing to advance life sciences, the possibilities for reducing time to market for medicines and devices and informing future investments are truly exciting.
Lowering patient recruitment and retention hurdles will make trials less time-consuming and expensive. With its support for health data interoperability, the Real-World Insights platform enables the use of research-grade evidence for synthetic control arms in clinical trials. A synthetic control arm uses real-world data collected from sources such as past clinical trials, natural history studies, electronic health records, claims data, disease registries, and smart devices. Without the need for a human control arm, all study participants receive the active treatment and no one gets a placebo. Imagine the potential for investigating rare diseases, where the lack of available patients make randomized clinical trials nearly impossible.
Decentralized trials can also increase patient participation. Through use of wearable devices, mobile apps, and telehealth check-ins with physicians, researchers can gather data while reducing the burden of onsite visits. The ability to integrate and share data securely from multiple physical sites—be they healthcare settings or patients’ homes—makes it easier for health providers and plans to collaborate with trial sponsors on patient recruitment and monitoring.

Next, consider an approved treatment or device that’s already in the market and shows promise for new uses. Based on real-world data collected during the product’s post-approval safety assessment, a new indication may arise, leading to the possibility of expanded label use. The 21st Century America Cures Act, enacted at the end of 2016, directed the FDA to evaluate and issue guidance on the use of real-world evidence to support approvals for new drug indications. This means that life sciences companies, as long as they have supporting data, can apply for expanded label use without having to conduct a new randomized clinical trial—a considerable savings of time and expense that directly benefits patients.
Another opportunity is longitudinal studies used to observe a product’s effectiveness and safety, which are more expensive to run than cross-sectional studies but offer a much better understanding of treatment patterns. Today’s connected devices offer intriguing options for collecting de-identified observational data over a long-term period. These datasets can be useful not only for monitoring treatment efficacy, but also for spotting public health trends and gaining other vital insights that can inform public policy and private sector planning and investments.
Additionally, researchers can use longitudinal studies to design future clinical trials and study treatment effectiveness. By offering pre-configured modules for collecting data from a multitude of sources, the Real-World Insights platform makes long-term data collection more cost effective, which supports patient and population insights during and after clinical trials.
Putting it all together
Standardized healthcare data formats and interoperability across many datasets over the course of time offer life sciences companies a wealth of information they can readily apply toward improving and saving lives.
The COVID-19 pandemic showed us how critically important real-world data is to understanding a disease and how to combat it effectively. Beyond trials, real world insights offer a path to understanding the whole person and their “-omics” (genomics, proteomics, etc), bringing us closer to personalized medicine and a new era of opportunity for life sciences.
With so many organizations still not fully satisfied with their real-world data platform, why continue to settle for something that doesn’t meet your needs?